When you view the image on our cover, do you see art or science? An exhibition at Princeton’s Friend Center — Art of Science — brings the two together. More than 160 images were submitted in the School of Engineering and Applied Science competition; 45 were selected for display through Reunions. The works spotlight the often-unexpected beauty of scientific findings.
Subjects range from extra-tropical cyclones to embryogenesis to the pathogen that causes cholera. “I was looking for images that are interesting in themselves but also reveal an interesting story in visual terms,” says James Steward, director of the Princeton University Art Museum, who judged the competition along with photographers Emmet Gowin, professor emeritus, and Jeffrey Whetstone, a visual-arts professor. “I particularly love the way in which this competition seems to spur scientists to visualize their work differently, and therefore perhaps gain different perspectives on it,” Steward says.
To view the full gallery of images online, go to http://bit.ly/artofsci2017
Images courtesy Art of Science Exhibition/Princeton University

Crystal Sunrise
Professor Lynn Loo and J. Clay Hamill Jr. GS
This image shows a single crystal of a perovskite material used for converting sunlight to electricity. Hamill dissolved two precursors — chemical compounds — in a common solution, then placed a drop of the resulting solution on a glass slide. As it dried, the precursors reacted to form this crystal, which is approximately the width of 1–3 human hairs; the streaks in the background are “much smaller perovskite crystals formed from precursors that dried before they could be incorporated into the large crystal,” he explains.
Loo notes that such a “stand-alone, perfect single crystal” is very difficult to grow, as they form only when conditions are “just right.”
Hamill says he was taken aback by the symmetry of the main crystal and the intricate patterns in the background: “I think it’s incredible that two precursor powders, under the right conditions, can form such a well-ordered structure.” He hopes observers of the image — which was taken with a microscope that uses electrons instead of light — will come away with an appreciation of minute objects and materials science. A close look “reveals the incredible ability of materials to order themselves when conditions are favorable,” he says — an ability that accounts for many materials we enjoy every day.

Face Warp
Ohad Fried *17
A postdoctoral fellow at Stanford who works at the intersection of computer graphics, computer vision, and computer-human interaction, Fried looks for new ways to think about photo and video editing. For example, he has come up with techniques to re-orient selfies to reduce distortion. This image of a human face was created as part of a project to enable sophisticated edits of portrait photos by rotating the face.
The image shows a warp-field: a representation of vectors that are used to “move pixels around,” Fried explains. Hue indicates direction, and saturation indicates magnitude. “When I first generated the image, I thought it was beautiful,” he says. He hopes viewers “find it interesting to see a human face represented in an unconventional way. There’s probably a deeper meaning hidden in that — I just don’t know what it is.”
For a video on Fried’s work, go to https://bit.ly/friedvid
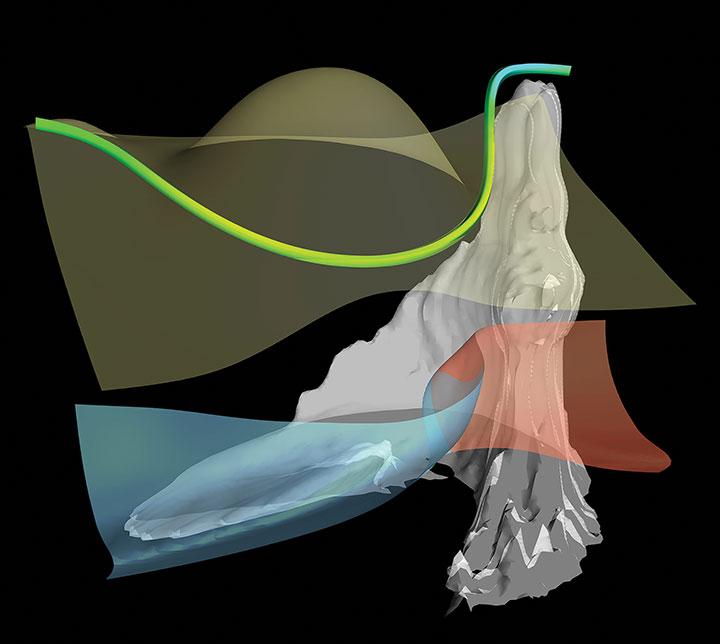
Clash of Air Masses
Tsung-Lin Hsieh GS
Among the topics studied by Hsieh is how clouds influence the intensity of extra-tropical storms. These storms form when warm air from the south mixes with cold air from the north. This image is a three-dimensional visualization of a simulated storm, with warm air moving north behind the red surface and cold air moving south behind the blue surface. The whitish surface represents clouds.
Hsieh wrote computer code to display elements of the storm, including clouds, wind, and temperature, and added colors and lighting to make the image easier to understand. He says he could look at the image all day long, because it “gave me a better intuition of what’s going on inside a storm and helped me develop a theory to explain my experiment results.”
When we see weather images in the news, he notes, we see them in two dimensions. “With computer-simulated data,” he says, “we would be able to see the fascinating 3-D structure of this giant system that affects our daily lives.”
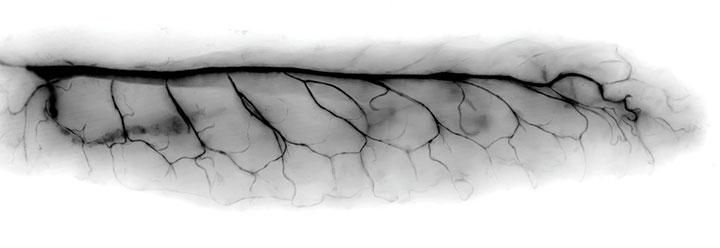
Breath of Life
Yogesh Goyal *17 and Heath Johnson, postdoctoral fellow
“Even a maggot is beautiful in the right light,” Johnson says of this image of the tracheal branches of a fly larva. Just before hatching, the larva takes its first “breath” as the trachea fills with air, contrasting it with the surrounding tissue.
Johnson and his collaborator, Goyal, stumbled upon the image as they were studying the formation of the tracheal branches, looking for defects. It was only after the researchers viewed the image again on the computer that they realized how beautiful it was, showing a repeated and precisely defined branching structure.
“Animal development is so complex, and it is remarkable that it goes right so often,” says Goyal. “This picture provides a sneak peek into the three hallmarks of animal development: robustness, reproducibility, and precision. In every developing larva, the highly branched patterns are produced without mistake in the right place and at the right time.”
To see more images by Goyal, go to https://bit.ly/2GSSrJ2

Bacterial Communication In Complex Geometry and Flow
Kevin Kim *17, Professor Howard Stone, Professor Bonnie Bassler
Bacteria use a chemical communication process known as quorum sensing to control collective behaviors including the formation of biofilms, which grow and adhere to surfaces. But the process does not take place uniformly. These researchers found that “bacterial colonization and biofilm development under flow and complex geometries can lead to heterogeneous quorum-sensing activation, which promotes diversity in the genetic programs that bacteria enact.” In this image, taken by Kim, the film grows on the surface but responds differently in the main channel than it does in the side channels.
Stone says he finds the general contrast and the differences between the biofilm in different parts of the image to be especially beautiful.
What should observers take from the image? To “appreciate the visual beauty that also can give insights into the mechanisms of different processes in science,” Stone says.

On the Cover
Octopus, October 21, 1952
Oriana Poindexter ’11
The octopus in the image on our cover was found by an unidentified scientist Oct. 21, 1952, in the lagoon at Bikini Atoll in the Marshall Islands. Poindexter, the photographer, found it 63 years later, hidden among millions of specimens in the Scripps Institution of Oceanography (SIO), where she received a master’s degree. She realized that the octopus was found at the time atomic bombs were being tested in the region.
“This set my imagination into a tailspin,” she writes. “Was this octopus radioactive? Why were there scientists out collecting octopuses at that point in time and space? What did they learn from their work there? … With some research, I learned the scientists were part of the Capricorn Expedition, the first SIO expedition to use scuba divers to explore the Pacific, focused on investigating the aftereffects of Operation Crossroads (the first nuclear detonations at Bikini Atoll in 1946) and overlapping with Operation Ivy, the first U.S. tests of a thermonuclear bomb in late 1952.”
Poindexter describes the SIO’s marine collections as “libraries of the sea.” As a photographer and scientist, she says, “I am fascinated by this combination of scientific potential and aesthetic beauty that these specimens represent.”
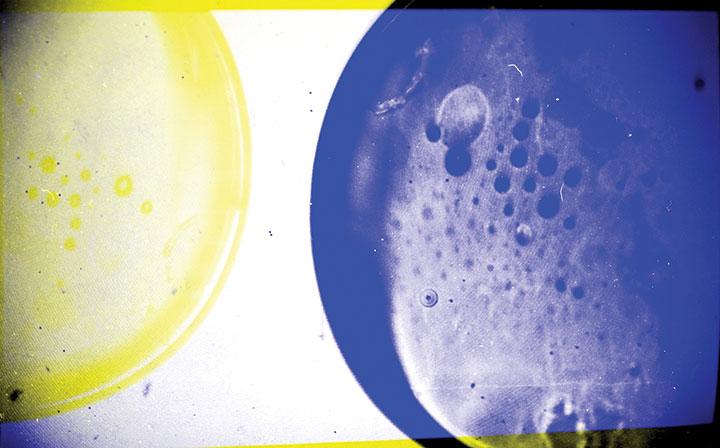
Bumper Moons #7
Louisa Willis ’17
Willis is an artist and studio assistant in New York City. Using 35mm black-and-white film, Willis shot photographs of petri dishes with red, green, and blue filters over her camera lens. Although the film is black and white, when the negatives are digitally scanned and layered on top of each other in the correct color channels in Photoshop, the original color image is restored. This replicates an archaic process, recalling the earliest color photographs. For this image, Willis turned that process inside out. Experimenting manually with colored lens filters, she then digitally layered the images in the “wrong” color channels, yielding an array of beautiful and unexpected colors.
Willis, who studied art at Princeton, learned about this process of creating color images from black-and-white film in a course with photographer James Welling, who encourages students to experiment with new techniques to produce images. He teaches a course called “Pathological Color.”
“I love how the images bear the traces of how they were made. If you look closely at the borders you’ll notice there is a layering of negatives here,” Willis says. “It gives the image a kinetic quality.”

Penguin Tracks
Michelle Greenfield ’18
In 2015, Greenfield was an intern at the South African Foundation for the Conservation of Coastal Birds, where she was responsible for feeding, medicating, and caring for rescued African penguins and other sea birds. One day she released 14 penguins back into the wild after a long rehabilitation process. “I was standing on the beach next to the line of penguins running back to the water, and I noticed the patterning of the tracks they were leaving in the sand,” she recalls. “I wanted to document their journey back home.”
Greenfield hopes the image will prompt observers to think about the hard work of animal-rescue teams and to recognize that all animals deserve study and appreciation.
“I think the entire process of rescue, rehabilitation, and release is beautiful,” she says. “It’s amazing how a few simple tracks can say so much about an animal’s life history.”

Crystalline Mondrian
Michael Fusella *17
This image shows a thin film, about one ten-thousandth the width of a human hair, of an organic molecule called rubrene. The film has been grown in such a way that the molecules have ordered themselves into a periodic lattice, forming a crystal. The different colors in the image identify individual rubrene crystallites. This crystalline thin film is used to enhance performance in electronic devices such as organic solar cells, organic LEDs, and organic transistors.
Fusella explains: “Typically, most organic thin films grow in a disordered (i.e., non-crystalline) fashion when deposited onto a conventional glass substrate. A large part of my doctoral research was to investigate practical methods to enhance thin-film crystallization of organic molecules. While it was difficult to tell by eye whether or not I was successful in making a high-quality crystalline film, the crystallites become easy to identify when imaged using a polarized optical microscope, which is just a conventional light microscope with two crossed polarizers in the light path. … This particular image is of a ‘patterned’ rubrene thin film, which means that the rubrene was only deposited in certain places on the substrate (and ultimately the crystals in this image were turned into an organic transistor!).”
As the image developed — the crystallites filling the rectangle with the black background around it — it reminded Fusella of work by abstract painter Piet Mondrian. “I hope the image convinces viewers that science isn’t all about hard data, abstract algorithms, and calculations; rather, there is much beauty in science as well,” Fusella says. “And if the image piques one’s curiosity to learn more about materials science or organic electronic devices, or even inspires a young person to pursue scientific study, it will have more than served its purpose.”
To view the full gallery of images online, go to http://bit.ly/artofsci2017


