FOR A LONG TIME, scientists believed that a mammal’s brain stopped adding neurons — the building blocks of the nervous system — soon after birth. When children, teenagers, or adults learned and formed new memories, it was thought, there were changes in the connections between existing neurons, not the creation of new ones.
That once-basic tenet of neuroscience has been toppled over the past few decades, and Elizabeth Gould, the Dorman T. Warren Professor of Psychology and Professor of Neuroscience, has been in the vanguard, thanks initially to her research on the growth of new neurons — neurogenesis — in adult rodent and primate brains. Her lab at Princeton has pushed beyond neurogenesis and now looks at how different experiences — from exercise to living in a given environment to stress to being obese — can change the brain’s function and structure in myriad ways. And while Gould’s work has not been in humans, it has implications for the more complex human brain, as well — possibly leading to an understanding of problems that can beset it, helping us to remember and learn better and to have healthier lives.
Gould didn’t set out to challenge scientific dogma. But as a postdoctoral student in Bruce McEwen’s lab at Rockefeller University, studying how hormones influence the brain, she noticed something curious. While performing an experiment that removed the adrenal glands of a rat, she observed a massive die-off of cells in the brain’s hippocampus, a region tied to memory formation and learning. But there wasn’t a net decrease in the number of cells, suggesting the dying neurons were being replaced. “I thought, ‘This isn’t happening,’” she remembers.
That was in 1991, so Gould’s search for an explanation took her elbow-deep into printed volumes of scientific literature. “I remember going to the Rockefeller library, and looking through these physical databases,” says Gould. She found that a handful of scientists had observed new neurons in adult mammalian brains as far back as the early 1960s, but their work had been ignored or attacked. She already knew about the work of Fernando Nottebohm, another Rockefeller professor, who had shown starting in the 1980s that the brains of adult songbirds living in naturalistic conditions generated new neurons. Encouraged by those precedents and by her own findings, Gould switched her research focus and persisted — even when conventional wisdom was that the whole idea was nonsense.
For a while, Gould’s work was ignored, too, recalls Heather Cameron, who studied neurogenesis with Gould at Rockefeller and is now a senior investigator at the National Institutes of Health. But Gould continued her research. “She rediscovered this phenomenon of neurogenesis that had been reported by several investigators and then pooh-poohed by others,” says her former adviser McEwen, a Rockefeller professor of neuroendocrinology who focuses on the effects of stress on the brain.
In 1997, Gould moved to Princeton. She and McEwen published papers showing adult neurogenesis in the hippocampus of the tree shrew (a small mammal) and the marmoset (a tree-dwelling primate). And in 1999, she and Charles Gross, now a professor emeritus at Princeton, described adult neurogenesis in several different types of monkeys, including macaques, that are more closely related to humans. In a paper published in Science that same year, she and Gross described neurogenesis in the cerebral cortex — the outer layer of the brain involved in higher thought, language, and other complex functions — of those monkeys. The findings were startling.
Over time, with more studies, neurogenesis in the primate hippocampus became generally accepted. Recent post-mortem studies by other researchers even suggest that middle-aged humans generate new neurons in the hippocampus. Neurogenesis in the cerebral cortex is still controversial; the issue is hard to settle in part because current lab techniques make it difficult to study.
Now, Gould’s lab is investigating more widespread forms of adult-brain plasticity — the brain’s ability to modify its own structure or function in response to changes in the environment of the body. For example, there are changes in the number, size, and shape of dendritic spines, which are like tiny antennae on a neuron that receive signals and transmit them to the neuron’s center. The number of synapses, the junctures where electrical and chemical signals pass between cells, also can change. In addition, her lab is investigating changes in glial cells, previously thought to not do much more than hold the brain together, but now suspected to be involved with cognition and other brain functions.
What sort of experiences — rewarding, stressful, enriching — produce these changes to the brain’s architecture? How do they relate to the way an animal behaves? Those are the questions Gould and her lab members are pursuing, with an ultimate goal of devising strategies to reverse cognitive or emotional problems that may be sparked by experience.
The lab uses rodents because their brains are good models, biologically and anatomically, for those of humans. But trying to connect a specific rodent experience to a human’s can be tough. A study published in 2013 by Gould and Erica Glasper, a former postdoctoral fellow, for example, found that middle-aged rats with sexual experience (sex is a rewarding experience for rats) will grow new neurons and see a restoration of some cognitive function. (It’s not known whether the neurogenesis actually causes the improvement.) That doesn’t mean 50-something humans should prescribe themselves constant sex for the sole purpose of boosting their brainpower — but it does raise the possibility that certain rewarding experiences for humans also might have cognitive benefits.
Running also is rewarding for rodents. Give a rat unlimited access to an exercise wheel, and he’ll spend many hours on it. Exercise has been shown to increase neurogenesis in rodents and to have many other positive health effects in both animals and people. Gould’s lab is trying to decipher the mechanics of exercise’s beneficial effects on the brain.
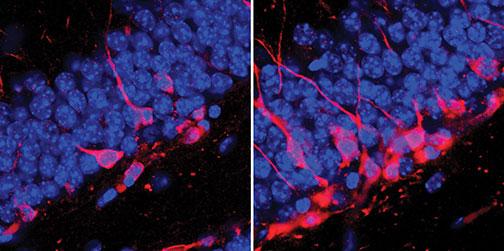
In a 2013 study, Gould and Timothy Schoenfeld *12 investigated a paradox of exercise: It reduces anxiety in both animals and people, but in animals, it’s been shown to spur the growth of neurons in the hippocampus that would seem to set the stage for anxiety. When mice were allowed to run as much as they wanted, their brains did produce many new, “excitatory” neurons — brain cells that activate other neurons they communicate with — more so than in sedentary mice. But the brains of the runners also showed an increase in activity in neurons known to inhibit other neurons. When the mice were exposed to cold water, the brains of the sedentary mice appeared to fire off those excitatory cells in response to the stressful experience. But in the running mice, the inhibitory neurons calmed the excitable ones. Exercise, the authors concluded, can insulate the brain from the anxiety associated with a stressful event.
A study presented at a meeting of the Society for Neuroscience in 2013 showed that rats allowed to run for 12 days better discriminated between objects in a task that depended on the medial prefrontal cortex — a part of the brain involved with object memory — compared to sedentary rats, explains graduate student Adam Brockett, who presented the work. The brains of the running rats also were different from sedentary ones, showing an increase in the number of dendritic spines and in the size of astrocytes (a type of glial cell) in the medial prefrontal cortex. So it appears that neurons are working with other types of brain cells to provide some of the cognitive benefits associated with exercise. “This highlights that brain functioning involves all cell types, not just neurons,” says Brockett.
Another major question for Gould’s lab: What do bare-bones surroundings mean for the brain, compared to those with more opportunities for stimulation? The former is the more common experience for lab animals. There’s evidence that rodents living in more enriched environments show increases in neurogenesis in the hippocampus as well as increases in the connections between neurons and in the number of glia, Gould says.
The same seems to go for primates. A 2005 study led by former Gould lab member Yevgenia Kozorovitskiy ’01 *07 compared the brains of marmosets who lived in a standard laboratory cage to those housed in larger cages filled with branches, straw nests, vegetation, and other objects of interest to monkeys. The brains of the animals living in the enriched environment showed structural changes that are typically tied to brain development.
More recently, Gould collaborated with Andrea Graham, an assistant professor in the Department of Ecology and Evolutionary Biology, to investigate an environment that was even closer to nature. In 2013, Graham adapted a special facility to study mice at the Stony Ford Center for Ecological Studies, a 99-acre parcel about 4 ½ miles from campus. Trailer-home-sized enclosures housed seven female rodents each and included “dirt and all kinds of New Jersey weeds and grasses,” says Graham. Mice were released into a miniature log cabin within each enclosure, and they had to learn to navigate the larger space and seek out the rodent chow and water provided by researchers over a three-week period.
As “clean” mice bred for the lab, “it had been tens of thousands of generations since their ancestors were outside,” Graham says. “It was sensory overload.” They eventually began behaving more like mice in the wild. Graham is interested in how the mice’s gut microbiome — the bacteria and other microbes that live in the digestive system — and thus the immune system are changed by the experience of living in a semi-natural environment.
When Gould heard about the proposed experiment, she realized it would be a great way to study the effects of that environment on the brain. After the experiment, she received the brains of the mice and found that “there was a massive increase in neurogenesis,” Gould says. The mice learned to navigate their surroundings, they burrowed, they scouted food — and it all added up to new neurons.
Life experience, of course, also includes how one relates to others, for example, in a social hierarchy. Gould’s lab has shown that the brains of dominant rodents show more neurogenesis in the hippocampus than subordinates. More recently, researchers looked at what happens when that hierarchy is shaken up. They effectively cast 12 rats into a season of something akin to “Real World: Rodentville.” Two separate groups of four males and two females living in the lab had their activities constantly recorded, even in darkness. The structure featured clear tunnels and the equivalent of an “open field” where they could burrow, “run around, chase each other, and engage in naturalistic behavior,” says Maya Opendak, a graduate student in the lab. Those conditions enabled the animals to form the sort of hierarchy seen in nature.
Once a dominant rat had established himself in each group, in about three days, they were swapped between communities. After the switch, the dominant rat lost his position and a subordinate took his place. But chaos reigned. “We didn’t see a decrease in fighting after four to six days,” says Opendak, who presented the study at the 2013 neuroscience meeting.
When the scientists studied the rats’ brains, they saw that the disruption suppressed the growth of new neurons in the hippocampus. As far as neurogenesis goes, the shakeup caused the dominant rats “to lose any benefits of being dominant,” says Opendak. And all of the animals had less neurogenesis than control animals that weren’t subject to the disruption.
Despite that, the disrupted rats didn’t seem to lose any function when put through their paces on tasks like social recognition and object location, which are dependent on the hippocampus. In fact, when tested on the rat version of anxiety, they were less anxious than controls, suggesting resilience, says Opendak. But when tested on social preference — basically, whether they’d rather hang out with a rat they’d previously met or a new one — they preferred the familiar, which may be an adaptive, “street-smart” behavior. That’s in contrast to the usual rat behavior, which is to spend more time with the newcomer, she says. Now researchers are trying to see whether suppressing neurogenesis without using social disruption as the mechanism produces the same behavior changes.
It’s a leap to map this work directly onto people, cautions Opendak. (Human social hierarchies are usually more subtle and complex, after all.) But one of the early takeaways for rats, at least, is that “even if you have some sort of disruption, it doesn’t have to be a life sentence.”
Still, Gould’s work also has suggested that certain disruptive events during a crucial window, in early life, may have much more lasting effects. A lab study published in 2004 showed that separating baby rats from their caregivers for extended periods suppressed neurogenesis far beyond the initial stressful event. When those rats were adults, even low levels of stress hormones seemed to suppress neurogenesis. This did not happen to adult rats that had not experienced early-life trauma. It’s not known whether reduced neurogenesis contributes to the ill effects of early-life stress seen in humans, but it may be one part of the puzzle.
The lab also is investigating obesity, which shrinks the brain and is associated with cognitive decline both in lab animals and in people. The shrinkage seems to come through the loss of synapses. “What we really want to understand are the mechanisms for why we see those reductions in brain volume and structural changes,” says Elise Cope, a postdoc in the lab who is leading the research.
The scientists are looking at the possible role of microglia, a type of glial cell that essentially acts as a central-nervous-system vulture. The cells scout the brain and spinal cord for damaged cells or synapses, infectious agents, or other debris, and then engulf or destroy them. (Recent research shows they’re also involved in sculpting neural circuits, by pruning weaker synapses in order to reinforce the stronger ones, says Cope.) In obesity, the microglia might be releasing inflammatory chemicals that cause neurons to degenerate. Or they might be prematurely engulfing synapses, and causing cognitive impairment that way. Cope is trying to find out whether those microglia actually cause synapse loss and cognitive decline. And if they do, can drugs inhibit the microglia and in turn prevent the decline?
In November, Gould’s team published a paper in The Journal of Neuroscience looking at the role played by electrical synapses — the electrical connections between neurons — in anxiety. The study showed that, in mice, blocking those electrical connections in certain parts of the hippocampus and prefrontal cortex decreased anxiety-like behavior. The connections seem to be important for anxiety regulation under some conditions. That raises the possibility of developing drugs targeting the connections as a way to treat anxiety disorders.
The work of Gould and her lab members is dedicated to the idea that the brain indeed can be changed, even in adulthood. What we do and what we experience matters to our brain’s very structure and function. And in turn, the keys to healing some forms of damage may also lie in experience.
“Behavior and experiences change the architecture of the brain,” says McEwen. And so stress and lifestyle factors such as sleep and exercise “are as important as our genes in terms of health,” he says. Gould’s work, he says, “has provided a foundation for understanding this.”
Katherine Hobson ’94 is a freelance writer specializing in health and science.


